
| Search our quarter-million Q&As |
Home of the finishing HOTLINE since 1989
-----Formula for electroless nickel plating solution
Q. Hi all,
Like a weirdly large number of other commenters in this thread, I'm a graduate student trying to make an electroless nickel bath for some small scale jobs. I worked in the plating industry for a couple years and you all saved my butt a few times (and made me look very competent despite being so green) so I figured this is the best place to ask.
I'm plating on plastic using ASTM B727-04 and the palladium/tin chloride seeding method. Our plastics are special proprietary blends but I've already done the legwork to develop the etching process.
My final plating solution is an alkaline sulphate EN bath that uses sodium hypophosphite as a reducing agent and tetrasodium pyrophosphate + triethanolamine as stabilizers (and pH control). It goes extremely slowly or not at all at temps around 40 °C but activates and plates beautifully at 50 °C. The problem is that it plates out extremely fast (<15 minutes). I'm trying to get 100 microns of plating so that's not going to work.
Before I start to play with the temperature and reducing agent concentrations, I was wondering if anybody had any insight on some additives or techniques that could help with this. I know the simple answer is to buy a proprietary solution, but I'd like to try with the chemicals I have before getting my boss to buy premade stuff.
Thanks everybody!
- Madison, Wisconsin
November 18, 2020
A. Hi Brandon
. All or most vendors substantially reformulated in just the last few years as lead and other non-RoHS stabilizers were removed. Still, probably nothing is more important than accurate temperature control because deposition rate is nearly asymptotic per Lowenheim's "Electroplating"
⇦[this on
eBay ,
Amazon,
AbeBooks affil links]
and "Modern Electroplating"
[on
AbeBooks,
eBay, or
Amazon affil links]
. And you must be careful not just to control it overall but to avoid hotspots. If you are able to get from 40 °C to 50 °C is less than say an hour, it's possible that local overheating is part of the problem.
Another thing to keep in mind when prototyping is that you may be tempted to use a tank which is too small for the surface area. Lowenheim says about 125 cm2/L is appropriate.
Luck & Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
⇩ Similar, related, Q&As -- oldest first ⇩
Q. I'd like to find out the exact recipe (components) for electroless nickel plating bath on steel (common, no special alloys). The layer has to ensure protection against corrosion. A layer about 8 to 10 microns is enough. Is somebody so kind to help me with this information?
Thank you in advance.
Nico Gaman- Nuenen, Holland
2002
2002
A. Hi Nico. Lowenheim's "Electroplating" offers over a dozen different formulations, and you'll probably find some additional formulas in other texts and literature from the 1950s, 1960s, and early 1970s, but electroless nickel formulas today are for all practical purposes proprietary. In other words, no user formulates them from basic compounds; they buy it from one of the many suppliers to the plating industry. That way you get a solution that works, you get full operating instructions (technical data sheet), you get analytical help, and you avoid mistakes like materials that are not RoHS-compliant .
You may already realize this, but plating jobshops are available which offer the service as well. Good luck.

Ted Mooney, P.E.
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
Multiple threads merged: please forgive chronology errors and repetition 🙂
Q. Hi-
I'm a graduate student in Physics at N.C. State University and I'm looking for a chemical formula for a electroless nickel plating bath. I'm working with a catalyst that is specific to phenethyltrimethoxysilane, non lead.
Thanks.
North Carolina state university - Raleigh, North Carolina
2005
|
|
A. I'll bet you would find many formulas for electroless nickel in a good university library...I assume NC State has a good library and a grad student can use it?  James Totter, CEF - Tallahassee, Florida 2005 A. You are in the realm of current technology and therefore most of the formulations are proprietary. You might try to research patent information for general information. Otherwise search the web for technical articles. Probably you will need to develop your own formula. process supplier - Great Neck, New York 2005 |
Multiple threads merged: please forgive chronology errors and repetition 🙂
Q. Hello,
My name is Maryam Kheiri. I am a university student and I am searching for the formula for electroless nickel, but I can't find it.
I need a complete procedure for using this method.
Thanks a lot
student - Iran
October 29, 2011
A. Hi Maryam. Almost surely your university or other local technical library will have a copy of either Lowenheim's "Modern Electroplating" or his "Electroplating". Each has more than a dozen formulations you can experiment with, plus excellent discussions on the effects of different variables. Good luck.
Luck & Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
July 2021
|
|
A. The manufacturers of electroless nickel plating solutions have spent many years and dollars developing the technology.  Jeffrey Holmes, CEF Spartanburg, South Carolina November 7, 2011 A. There are many books on that subject, in fact any better contemporary plating handbook must have chapter on electroless plating. If you speak russian there is russian website where you can download (free) some old old ex-USSR books on that subject (www.galvanicrus.ru/). Hope it helps and good luck! Goran Budija- Cerovski vrh Croatia November 7, 2011 |
A. Obviously there are basic (generic) formulations for those baths they can be found in literature ASM handbook #5, Modern Electroplating by Schlesinger & Paunovic, and especially there is a book dedicated to that type of bath is on this site on the "must have" books list.
The best idea is to buy them not only for the authors dedication to develop these tasks, but also because it needs a wider understanding of the process equipment which is not covered having the formulation.
But if that is not possible please let me know and I will send you some formulations for them.
- Bucaramanga, Santander, Colombia
December 11, 2011
December 18, 2011
A. In this system you need nickel sulphate, sodium hypophosphite, and additives
For example :
100 lts bath make up, you need:
------------------------
3 kgs nickel sulphate
3.750 kgs sodium hypophosphite
6 kg additive
Is this enough from me?
regards
- Istanbul, Turkey
Q. Thanks for your help, but as you know I live in Iran. I can't get information so easy and fast and I really need a complete procedure. I ask some people here but they sell their information very expensively and I can't pay for them.
So I need your help, thank you/
- Iran
December 24, 2011
December 30, 2011
A. Mr/Mrs
Electroless nickel has several steps.
But before that:
What kind of substrate is the one you want to plate?
If it is plastic it needs an etching step which is done in a concentrated solution of chromic acid plus sulfuric acid
It works fine for ABS plastic but the other types of plastic I do not know; literature says PVC and others can be plated ok, but the best is ABS.
And then the surface must be rendered conductive.
If it is a conductor, well here are basic bath formulations --
acid baths using sodium hypophosphite
1)
nickel chloride 30 gr/litre
sodium hypophosphite 10 gr/litre
hydroxyacetic acid
⇦ on
eBay
or
Amazon [affil link] 35 gr/litre
pH 4-6
temperature 100 °C
2)
nickel chloride 30 gr/litre
sodium hypophosphite 10gr/liter
sodium citrate 12.6 gr/liter
sodium acetate 5 gr/liter
pH 4-6
temperature 100 °C
There also are alkaline baths
Also you can find different reducing agents like sodium borohydride, and dimethylamine borane and hydrazine.
- Bucaramanga, Santander, Colombia
A. nickel sulphate 40 g/l
sodium hypophosphite 26 g/l
succinic acid 50 g/l
temp 85-95 °C
- Bucaramanga Santander Colombia
January 27, 2012
Q. Dear sir
What is the ratio of electroless nickel in sodium succanate, sodium citrate, hypo, nickel salt per lt?
And how to make activator with silver?

Sunil Jain
- Mumbai, India
February 5, 2015
A. Hi Sunil. We appended your inquiry to this thread where some of the answers you seek are already available. Good luck.
But please provide us the background of who you are and what you are trying to do because otherwise the answers provided can be misleading. For example, in general, individuals don't formulate such solutions from raw chemicals, they buy them as proprietaries -- but we don't know if you already know that.
If you are a PhD student though, you may want to play with these solutions in the lab anyway as a matter of research. If you are a plating shop, most of us would consider it wasteful to make expensive solutions that will not yield enough turnovers because they do not contain the carefully developed proprietaries.
Silver based metallizing solutions based on the Brashear process and similar mixes of silver salts and reducing agents can perhaps be used for your application (you haven't told us what it is), but it may be a poor idea and it is not the most common route to metallizing materials for subsequent electroless nickel deposition. Best of luck.
Regards,

Ted Mooney, P.E.
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
February 2015
Q. I'm manufacturing gold/silver plated idols & statues on fiber cultured marble

Sunil Jain
- Mumbai, India
July 2, 2021
A. Hi Sunil. David Hernandez gives his formula for generic electroless nickel using those ingredients just above in his posting of Dec. 30, 2011. A good explanation of two-part silvering, and a discussion of how to sensitize the surface for it is in thread 23536. Good luck.
Luck & Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
July 2021
Q. Is there some sequences in adding constituents (stabilizers, complexing agent, ...) in order to make electroless nickel bath?
Mohammad rezaTUW university - Vienna, Austria
October 16, 2015
October 17, 2015
A. Hi Mohammad,
The rule of thumb is to add everything first (reducing agent, pH buffer, stabilizer, etc.) prior to the nickel sulphate.
Regards,
David

David Shiu
- Singapore
Q. Dear Sir,
I am a Chemical Engineering student and I have to do a project on electroless nickel plating. I would like to know if there is a formula for a medium nickel-phosphorus solution in order to plate 4140 steel. I have not decided on a thickness yet.
Could you please let me know what are the proportions of the components of the bath and the amount of area this quantity is able to plate?
If anyone could help me I would really appreciate it as I am a student and can't afford to buy this information from a supplier in the industry.
Louisiana State University - Baton Rouge, Louisiana, United States
July 7, 2017
July 2017
A. Hi Maria. Although there are technologies that are "licensed" such that you buy the knowledge, and there are also consultants who can sell you the knowledge, in electroless nickel plating you generally "buy the process", i.e., the seller provides their products by the gallon, with the necessary instructions.
But if you still want to make your own solution, start with any of the formulations shown on this page, and ask detailed followup questions if necessary regarding the answers already offered. The substrate being 4140 steel probably doesn't affect the formulation. Alternately your university or city surely has books on the subject. Lowenheim's "Modern Electroplating, for example, offers 8 formulations for hypophosphite baths, plus 4 formulations for borohydride baths, and 6 for DMAB. Good luck.
As for how long the bath will work, understand that however much nickel you want to put on the parts had to come from the bath. So if you are doing a generic formula capable of only "one metal turn-over", the amount of nickel in the make-up tells you the maximum amount of nickel you could possibly plate out from it. But, in Lowenheim again, is the claim that not more than a g-atom of nickel can be generated for each mole of hydrogen released -- so it's quite limited. Have a good project!
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
July 8, 2017
A. Hi Maria
Electroless nickel processes are not as simple as the published formulations suggest. You could waste a lot of time and money on chemicals and detract from your main project.
I would suggest that you approach the suppliers of established processes and explain your situation.
You will probably find them very helpful. Remember that today's poor student could be tomorrow's major buyer

Geoff Smith
Hampshire, England
Q. I am trying to make an electroless nickel plating bath.
composition of the bath:
Nickel sulphate, sodium hypophosphite plus C2H5NO2 as an Accelerator and C6H5NA3O7 as complexer and CH4N2S as stabilizer. as soon as I add the sodium hypophosphite to nickel sulphate solution there is precipitation which is insoluble in water.
Any idea about this issue?
Your advice is highly appreciated.
- Austin,Texas, USA
June 18, 2019
June 30, 2019
A. I had the same experience, I sourced the sodium hypophosphite from abroad because it's very hard to get in the USA (it's a controlled substance used in the synthesis of illegal drugs) and I got the same result as you did; I gave up trying to make my own formula. The good news is that there are USA-based providers that sell the electroless nickel plating solutions separately (Solutions A, B and C), the solution A is just Nickel sulphate (20 grams per litter of Di water), the solution B is what have the hypophosphite and buffers, the solution C is a rejuvenator.
For a Proof of Concept I just got the solution B and got good results from it (nothing commercial grade of course).. Just search for Electroless Concentrate Part B and you will find it easy, I have tested the same solution with Nickel sulphate, Cobalt sulphate, Iron sulphate, and other metal sulphates mixed with nickel/cobalt and the results are very promising for many types of industries.
A few pics of the chemicals and solutions I've got:
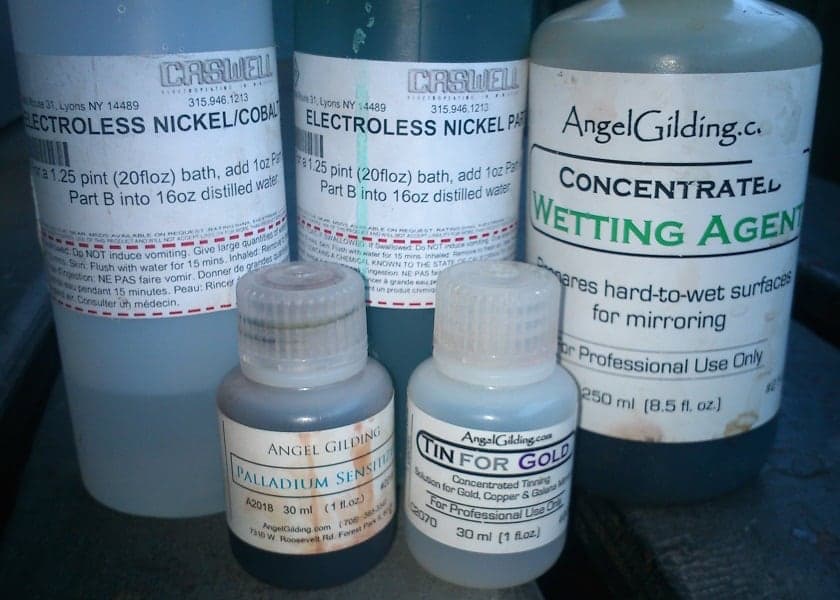

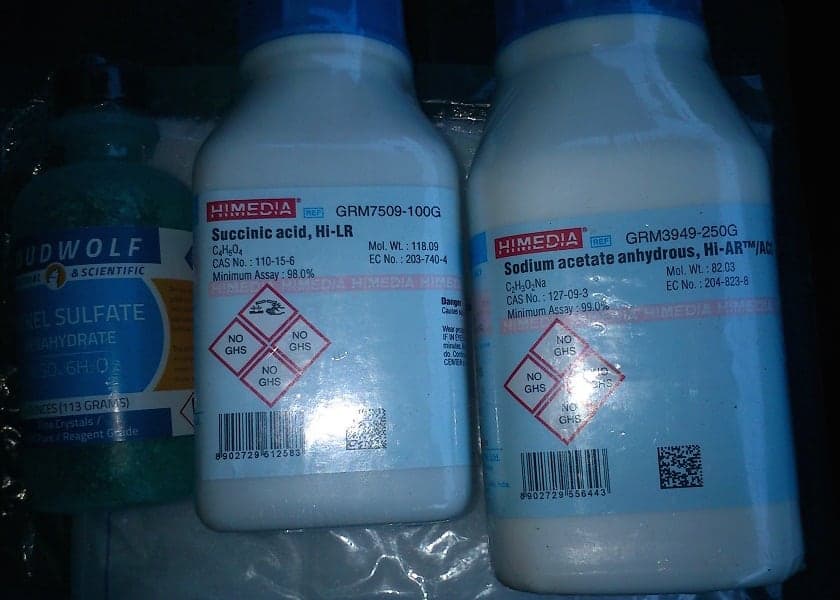
- Managua Nicaragua
|
|
P.S.: Mr. Mooney, perhaps the showing the pics is against the rules, if so, would leaving the links be fine? July 2019
Regards,  Ted Mooney, P.E. RET Striving to live Aloha finishing.com - Pine Beach, New Jersey Need quick confidential answers? $25 Need project assistance? $100/hr. July 2, 2019 Thanks Mr. Mooney.
- Managua, Nicaragua |
A. ASTM B656 will give you formulas for Autocatalytic Ni-P deposition.
Jim fultonA-Labcorp - Dayton, Ohio
December 16, 2019
Ed. notes: ASTM says that ASTM B656 referenced in Jim's posting is "inactive", but copies are available.
Sorry! Finishing.com is temporarily Read-Only.
Ted Mooney is retiring but I have several offers to take it over.
We're working hard to make sure we find it the best new home.


