
Curated with aloha by
Ted Mooney, P.E. RET

Plating -- How It Works
An introduction for students, explaining the principles and offering demonstrations
What is electroplating?
Electroplating (often just called "plating") is the deposition of a metal coating onto an object by putting a negative charge on it and putting it into a solution which contains a metal salt. The metal salt contains positively charged metal ions which are attracted to the negatively charged object and are "reduced" to metallic form upon it.
How does plating work?
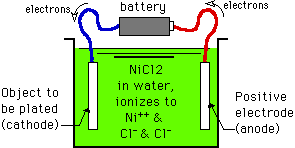
Look at the figure above: We have a metal object that we want to plate with another metal. First we fill a "cell" (a tank, vat, or bowl) with a solution of a salt of the metal to be plated. Most of the time the salt (nickel chloride in our example) is simply dissolved in water and maybe a little acid.
In this example, the NiCl2 salt ionizes in the water into Ni++ ions and two parts of Cl- ions.
A wire is attached to the object, and the other end of the wire is attached to the negative pole of a battery (with the blue wire in this picture) and the object is immersed in the cell. A rod made of nickel is connected to the positive pole of the battery with the red wire and immersed in the cell. The battery is pulling electrons away from the nickel anode (through the red wire) and pumping them over to the object to be plated (through the blue wire)
Because the object to be plated is negatively charged (by being connected to the negative pole of the battery and having electrons pumped to it), it attracts the positively charged Ni++ ions that are floating around in the solution. These Ni++ ions reach the object, and electrons flow from the object to the Ni++ ions. For each ion of Ni++, 2 electrons are required to neutralize its positive charge and "reduce" it to an atom of Ni0 metal. Thus, the amount of metal that deposits is directly proportional to the number of electrons that the battery provides.
Note: This proportional relationship is a reflection of Faraday's Law of Electrolysis. If you are advanced enough in chemistry (a high school student), that you've heard terms like gram molecular weight, mole, valence and Avagadro's number, but it's all a hodgepodge to you instead of a cohesive whole, don't despair! Study Faraday's Law for a while, which says that 96,485 coulombs of electricity will deposit one gram equivalent weight of metal, and all of these disparate wacky terms will come together in a moment of enlightenment.
Is this a little too advanced for your grade? Maybe What juice cleans pennies best? would be a better project for you?
Meanwhile back at the anode, electrons are being removed from the Nickel metal, "oxidizing" it to the Ni++ state. Thus the nickel anode metal dissolves as Ni++ into the solution, supplying replacement nickel for that which has been plated out, and we retain a solution of nickel chloride in the cell.
As long as the battery doesn't go dead, nickel continues to dissolve from the anode and plate out onto the cathode. As we move the electrons from the anode to the cathode, through the wires, and powered by the battery, the rest of the nickel follows, dissolving into solution at the anode, migrating through the solution, and plating out at the cathode.
Note: We picked nickel chloride in the example chiefly for simplicity of explanation. First, because nickel always dissolves in the "+2" oxidation state (Ni++), whereas many other metals like copper and zinc can dissolve in either the "+1" or "+2" state and add some confusion; secondly because chloride is a simple one-atom anion whereas many anions like sulphate or acetate are more complex. But we do not actually recommend that nickel be used for your first school science demonstration because -- while the explaining is simple -- the plating is difficult :-)
For school demonstrations, we suggest plating copper pennies with zinc, or plating quarters or brass keys with copper because both are easy and can be done without the need for any dangerous or toxic chemicals.
The first demonstration -- Plating a copper penny with Zinc
For the first demonstration, the cathode will be copper (pennies), the anode will be zinc, and the "electrolyte" (the solution) will be zinc dissolved in vinegar ⇦ in bulk on eBay or Amazon [affil link] and water.
Zinc anodes are available from boating stores ![]()
With a hacksaw a teacher or the group can cut many slices from one anode.
A second option to obtain zinc is to sand down a modern U.S. penny (1983 or later) until the copper surface is removed and the underlying zinc core is exposed. This source of zinc lacks some elegance, though, because it removes a little bit of the magic by showing that the penny was already electroplated, and it tends to be confusing, with some pennies zinc colored because of sanding before you even start the plating.
Note: A third possible source of zinc is the shell of conventional carbon-zinc batteries (make sure not to use alkaline batteries like Duracell or Eveready Energizers, nor rechargeable nickel-cadmium batteries -- just the cheapest 1-1/2 volt AA, C, or D plain carbon-zinc batteries). The science teacher can cut up such batteries and remove the black glop, and give the student the cleaned zinc strip.
For the pennies that you wish to plate onto, although any pennies will do, if you start with a dull brown penny, you'll end up with a dull zinc plated penny. Try to find shiny new pennies for best results! Immediately before plating, clean the penny with toothbrush and toothpaste; an even better cleaner, if you have it, would be Bon-Ami [link is to info/product at Amazon]. Rinse well after cleaning. Your hands are oily, so wear plastic gloves so you do not get fingerprints or other soils on the penny after you clean it.
A transparent plating container is best, a Pyrex beaker is excellent, but a glass dessert bowl can serve well.
A recipe for the solution, suggested by Tom Pullizzi, and found to work is:
1). Fill the container about half way with vinegar (vinegar is mild acetic acid).
2). Put the zinc anode into vinegar and let it sit for several hours, allowing some of the zinc to dissolve.
3) Add 100 g/l of Epsom Salt [link is to info/product at Amazon] (this makes the solution conductive; table salt will work fine, too, but is a little less safe because if students get carried away and start using really high voltages, table salt can generate chlorine gas, whereas Epsom Salt, magnesium sulphate, can't).
4) Add 120 g/l of table sugar. This is called a "brightener", and its function is to sort of interfere with crystal growth, causing more and smaller crystals for a brighter, harder, plating.
5). Connect one flashlight battery (1-1/2 volt D-cell) to the penny and the zinc anode, and place them into the solution. Don't let them touch each other.
With luck, within a few minutes you'll begin to get a bright silvery coating. Ted Mooney didn't have quite that much luck when he tried it, but did find that a reapplication of the toothbrush and Bon-Ami quickly polished the thin greyish coating he got to a fairly bright shine.
A second demonstration -- Copper plating a key or a quarter
Another slightly harder demonstration is plating a quarter or a brass key with copper. The key on the left was copper plated from a solution of vinegar with a pinch of table salt and a pinch of sugar, again using a 1-1/2 volt flashlight battery for power.

Understanding why this experiment is a little harder to do than the first one is a good science lesson: You can't plate a metal out of a solution until you can get that metal dissolved into the solution as ions (actually, this is why we didn't do a student demo of nickel, silver, gold, or chrome plating; you won't be able to dissolve these metals in vinegar, you would need a stronger and more dangerous acid),.
Copper will not dissolve in vinegar without electricity to help it along, so it's best to get started with a small piece of scrap as your cathode and a large coil of copper wire as the anode. I stripped and scrunched together about 2 foot of 14 gauge wire to use as the anode (wire is very pure copper) and used a 1/2 inch length of stripped copper wire as the scrap cathode. After I ran it this way for a couple of hours the solution acquired a faint blue tinge to it -- indicating that a little copper was dissolved in it. Then I cut off the scrap length of cathode wire, attached the key and plated it for several hours. vinegar is too weak an acid to hold much copper in solution, so there is no rushing it, you have to plate slow and for a long time so copper can slowly dissolve into solution to replace what you plate out. I found that just a pinch of table salt (maybe two shakes) was enough. If you use more, what happens is you make a more conductive solution, so more electricity flows, but since there is not enough copper dissolved in the solution to support that current flow, you generate a lot of hydrogen gas and deposit a lot of black "smut" -- you can't plate copper out of solution faster than it goes into solution!
What happens if your solution is too conductive due to too much table salt? The electricity is flowing through the solution, so electrons are flowing into it from the cathode. But if there are no copper ions there to pull out of solution, the electricity will pull hydrogen ions out of the water per this equation:
2H20 --> H2++ + 2OH-
This will cause bubbles of hydrogen gas to accumulate on your key or quarter, and the OH, the hydroxide, will neutralize your vinegar so you'll have no acid left.
What is "smut", or as some students call it, "black glop that coats the coin"? When you have too much current flow, what also happens is the moment that an ion of copper gets to the cathode, it is "reduced" instantaneously with no opportunity for proper crystal growth, and it forms a powder of tiny, non adherent individual specs of metal which appear black because they scatter all the light, reflecting none. So use 1-1/2 volts maximum, very little salt, and take your time!
The Experiment is done: now, Waste Disposal!
In the "real world" waste disposal is a truly major headache for plating factories. But here you have only used toothpaste, vinegar, salt and sugar. The small amount of zinc or copper you have dissolved into it is really not a significant issue. Still, we should learn to practice environmental responsibility, rather than learning to talk ourselves out of it! So, with the wires disconnected, put a good size chunk of oil-free and soap-free steel wool into your bowl of plating solution. The copper, and much of the zinc, will "immersion deposit", plating out onto the steel wool, thereby removing the waste from the solution. Copper metal is not a pollutant, but dissolved copper definitely is.
Postscript: This is about science principals, not robust electroplating!
This presentation has drawn people over the years to write to us, telling us about the parts they want to plate and asking what tweaks they need for heavier, more-decorative, more adherent, more robust, corrosion-resistant, functional plating -- and our answer has to be along the lines of: "What tweaks does a Wright brothers' airplane need for it to carry a passenger from New York to Paris?"
These experiments demonstrate the theory involved in moving metal from the anode to the cathode, but they skip all of the hard work of functional plating which is 99% of the job. Our FAQ "Introduction to Chrome Plating" explains some of that stuff. Some types of hobby plating are possible, but it is a subject for many pages of reading, lots of experimentation, training in handling hazardous materials, and the purchase of equipment and proprietary plating solutions. The short answer is that if you need something plated, sending it to a plating shop is usually the best answer :-) Good luck!
Please ask specific questions on our Hotline-Letters page! Save time and lost motion by mentioning that you've already read this page. Good luck!

