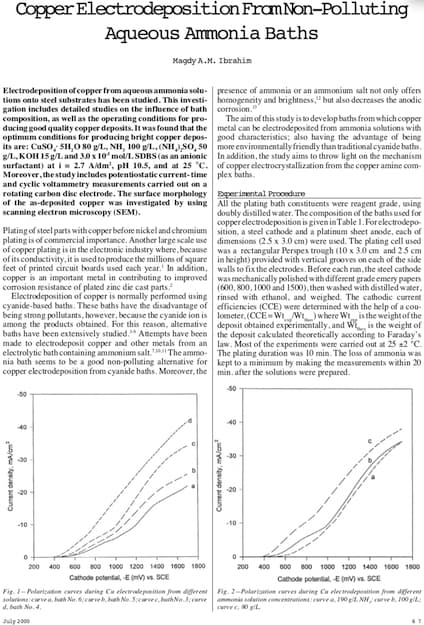
| Search our quarter-million Q&As |
Home of the finishing HOTLINE since 1989
-----Copper Plating from an ammonia Solution
Q. Hello I'm Jordan,
I have been doing electroplating as a amateur hobby for a while now and I have a question. I've tried looking up the answer but either I am not wording it right or I have overlooked it. I have made an aqueous solution of copper and ammonia. Would I be able to use it as is as a solution for copper plating? And if no, is there a way I can neutralize the ammonia
⇦ on
eBay or
Amazon [affil link] closer to a safe pH thats usable? What answer am I not thinking of or overthinking? And I am an hvacr tech so my shop does have a hood fan in it so vapors are handled in a safe manner. Thank you.
- Centralia, Illinois
January 30, 2021
A. Hi Jordan. I'm not familiar with any industrial copper electroplating being done from an ammonia solution, but apparently it is possible ==>
But I suspect that chloride or sulphate (as per the article) is required for conductivity if for nothing else. So where did you find instruction to try ammonia as the electrolyte, and why are you trying to do this? If you're just mixing random stuff together, an explosion is probably at least as likely as a workable electroplating solution :-)
Please tell us what motivates your efforts on this so we can help more effectively.
Luck & Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
A. Add some tartaric acid
⇦ on
eBay
or
Amazon [affil link] and water, pH must be 8-9, and then it must work. Alkaline tartarate bath is one of the oldest cyanide free coper plating baths.
Hope it helps and good luck!
- Zagreb Croatia
Sorry! Finishing.com is temporarily Read-Only.
Ted Mooney is retiring but I have several offers to take it over.
We're working hard to make sure we find it the best new home.