
| Search our quarter-million Q&As |
Home of the finishing HOTLINE since 1989
-----Energy savings in high temperature Type II anodizing
Q. As you know, normally temperature is 19-20 °C [66-68 °F] for type 2 anodizing. If I increase this temperature to 28-29 °C [82-84 °F] with proprietary/special chemicals (sodium bisulphate and others), then what will be energy savings as percentage if I compare with 20 °C [68 °F]?
Alaattin tuna- TURKEY,sakarya
April 18, 2020
A. Hi Alaattin.
We would need to know the room temperature & relative humidity, the tank dimensions, any insulation applied to the tank & the whole chiller system, whether you operate 24 hours a day, the rectifier operating amperage & voltage, etc., to do any calculations ...
But I think the short answer is "negligible" if you make no changes in your physical plant, but "significant" if you do.
The reason I say "negligible" is because the energy required to keep a tank at 19-20 °C when you are not operating it is negligible, and all of the heat put into the solution by the rectifiers must be removed either by casual loss to the environment or by your cooling system ... and the amount of casual heat lost to the environment by operating at 28-29 °C is rather negligible as well.
However, if the higher temperature allowed you to switch your cooling system from a chiller to direct operation from an evaporative condenser or a cooling tower, they use far less energy (you only need to pump the water, not to mechanically chill it).
Please look into whether there are possible advantages to higher temperature anodizing, not as a cost-saver.
On a personal note, my career in metal finishing was interrupted by a stint in the manufacturing technology division of a world famous consulting organization, you all know its name ... but I disliked my time there. One reason was that I was involved in "cost reduction" programs and never saw a single one I considered successful; my personal opinion is that the moment you shift focus from doing things better to doing things cheaper, you've taken your eye off the ball and the game is lost 🙂
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
A. Do not do it. As said above, negligible, because it will take slightly less voltage to get the same amps,. resulting in lower watts, but, the pore will be larger and will not seal and if dyed the dye will bleed.

Robert H Probert
Robert H Probert Technical Services
Garner, North Carolina

April 18, 2020
Q. My aim is just only chiller unit's less working ... less energy consumption means greener production and less carbon footprint and less energy cost, Right? So if I get this project with these special chemicals why not continue anodizing production by this system? Of course I will test anodizing quality of profiles by weight loss test in laboratory. I will evaluate all results at end of the project ... I think it's worth trying!
Alaattin tuna [returning]- TURKEY,sakarya
April 21, 2020
A. Hi again. I've already expressed my belief that the energy saving will be negligible, Alaattin. But I've been wrong before, so please show us some before-&-after energy logs when you've tried it. Thanks!
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
April 2020
A. The heat build-up in an anodizing bath is from the amps & volts supplied by the rectifier. In my opinion, you will spend more energy trying to figure out any savings then you will actually get. Both Ted and Bob hit the nail on the head "NEGLIGIBLE". Use your time to look into other areas where savings can be made like "Aluminum Cathodes"

Drew Nosti, CEF
Anodize USA - Ladson, South Carolina
April 23, 2020
June 4, 2020
Q. Hi Ted, How are you? I hope there is not violent protest in your location and you are ok. I want to share with you a calculation method which is related to my query.
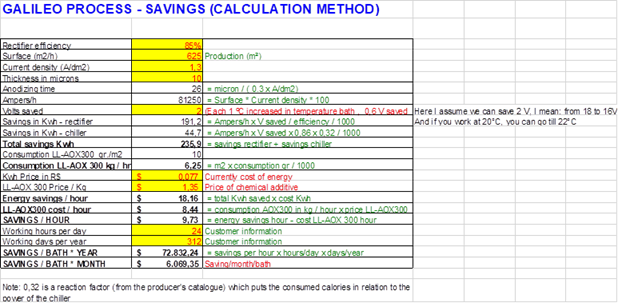
So, my question here is: - Could you check calculation? Is there any mistake with it? -Could you publish this chart on finishing.com as my answer to contributors? Alaattin tuna [returning]
- TURKEY,sakarya
A. Hi Alaattin. I can just barely read your chart and it's very hard on the eyes, so if you can send a larger one or higher resolution one that would be great.
I'm not going to decipher the math, but if 2 volts can be saved with no downside it will indeed save a lot of electricity. That, rather than the operating temperature is where the savings would come from, and that's the claim/assumption which you would want to see evidence for. Good luck.
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
June 2020
A. Alaattin
Using the supplied data...
Amperage is 81,250 amps per load,
Ampere hours is 35,208 (81,250/26 minutes) per load; savings will be overstated by more than 50%,
@ 1 load per hour, 4,680,000 square meters processed each year; double that if you have 2 hoists,
$/kwh appears to be off by a factor of 10.
- Green Mountain Falls, Colorado
June 5, 2020
Q. Hi again,
I have been testing a project for one week where my aim is energy saving, either electricity and chiller by adding oxalic acid to anodizing bath. I have added 10 gr/lt oxalic acid (it will change pore structure) to anodizing bath and I have increased temperature to 30 °C (86 °F) for that anodizing bath, and also I have decreased the voltage of rectifier.
When I checked the anodized profiles I did not see any negative appearance on profiles. I have checked coating thickness, dye spot test, even mass loss test were ok. There was not any white dust due to high temperature effect. As logically,I assume I have saved energy either electricity and chiller aspect.
So how can we evaluate these consequences?
Is there any mistake with my testing?
Is there anyone who object this project as technical aspect?
- TURKEY Sakarya
September 25, 2020
A. Hi again. The Watts of power required from the rectifier, which power must also be removed by the chiller, is the product of Amps x Volts. So the hypothesis that seemed worthwhile to me was the claim that the voltage could be reduced by 2 volts due to the higher temperature. Have you done any resistivity/conductivity test -- Haring Cell, or even as simple as measuring how many volts are required to maintain a given current for the two different operating conditions?
If the conductivity of the solution is so much higher that the reduction in necessary voltage is actually 2 volts less, then substantial energy savings sound possible. If the reduction in necessary voltage is not actually significant, but was just a baseless guess, this project strikes me as wasted effort. I think it all hinges on the truth of the claim that the conductivity of the solution is much higher.
Luck & Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
September 2020
Q. Hi Again. I think you are right Ted.This project seems like vain for energy saving issue aspect. I have tested 3 anodized bars which each of put different anodizing bath (one of baths is high temperature and oxalic acid added, other two are normal, 20 °C).Each of bars all same as pieces and square meter for three baths. Unfortunately, all currents were nearly same during anodizing. So ,your thesis which you claimed due to conductivity effect current was increased by temperature. So all three baths consumed same energy even if one bath is high temperature/low voltage. But should we think same issue as well as cooling energy consumption aspect? Or can we think chiller unit will be less loaded run on 20 °C instead of 30 °C degree?
Sincerely
- TURKEY Sakarya
September 28, 2020
A. Hi. If the voltage is actually significantly lower, the power is significantly lower, because Power = Current x Voltage.
The operating temperature really doesn't much affect the power required to cool the bath because the amount of heat lost to the surroundings or gained from the surroundings at 20-30 °C is negligible. What is the average temperature in the shop? -- I'd guess it varies from about 20-30 ° C 🙂
Luck & Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
September 2020
Q. Hi. Frankly, I do not understand how operating temperature really doesn't much affect the power required to cool the bath? We know anodizing process is exothermal reaction. That means during reaction (anodizing) heat comes out. This reaction is independent from surrounding, right? So to me, keeping bath temperature 30°C during anodizing something else and keep of bath temperature 20 °C something else.
So there is two different issue and both of conditions give different energy load for chiller unit.
Alaattin tuna- TURKEY Sakarya
[returning]
September 29, 2020
A. You've demonstrated by dozens of postings here over the years that you're both knowledgable and smart, Alaattin, so I'm not sure why we're not on the same wavelength 🙂
Nevertheless: the amount of heat you put INTO this 'exothermic' process is measured in Watt-hours, and is simply equal to Volts x Amperes X Hours of anodizing time.
If the temperature of the bath is close enough to room temperature that no significant amount of heat is going into the tank from the room, or leaving the tank and going into the room, then the amount of heat which must be removed by the cooling system is the same as the amount that went in:
Volts x Amperes X Hours of anodizing time.
If you were comparing two solutions which operated at 0 °C and 95 °C there would be a big difference because the 0 °C tank would absorb measurable heat from the room, and the 95 °C tank would lose quite a lot of heat to the room. I am claiming that the amount of heat gained or lost to the room at 20 °C vs. 30 °C is probably negligible.
Luck & Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
September 2020
Q. Thank you for your compliment Ted. I understand your approach to chiller energy subject. Also can we say volt*amperes*hours all equal for either 20 °C and 30 °C because of conductivity effect? (I think chiller capacity calculation was like this: volt*ampere*3,412=btu/hour).
I tested two bar in different temperatures in last week. I saw both bars were nearly equal ampere during anodizing. So volt*ampere value is same for both bars that means cooling effect same even if different temperatures. Did I explain properly or wrong?
Sincerely
- TURKEY Sakarya
September 30, 2020
A. Hi. Yes, if the Volts, the Amperes, and the Hours for both bars were all the same the heat input to the chiller is the same.
Luck & Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
September 2020
Q. Hi Ted,
I discussed this energy saving issue with my friend last week. I have said to him "energy saving for electrical aspect" is "NEGLIGIBLE" as you said due to "conductivity of bath". I have told him my tests. There were two bars (same profile, same piece) one of the bars was anodized in 77 °F and 16 V; other one was anodized 68 °F and 18 V. I have surveyed both bars required nearly same amperage on rectifier display. So I said in this conditions (equal amperage even if different voltage) we don't mention energy saving ... but he said we can mention; he has calculated total energy of both bars as Voltage * Amperage. He claimed low voltage bar's energy necessity (16 volt*5000 amp=80 kW) lower then high voltage bar's (18 volt*5000 amp=90 kW)
So, can be his approach true?
He confused me with this calculation
- TURKEY Sakarya
December 4, 2020
A. Hi again. We are apparently having language difficulties because I certainly did not say what you think I said 🙂
I said: "If the voltage is actually significantly lower, the power is significantly lower, because Power = Current x Voltage.".
Luck & Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
December 2020
Q. Hi again. Of course I know that formula which you had written but unfortunately I did not understand properly "effect of increasing anodizing bath's conductivity by temperature over electric energy saving". Because increasing conductivity means increasing current so if we compare 16V-77 °F and 18V-68 °F anodizing conditions, we will see same amperage for both condition due to conductivity effect. As if we can't mention any electric energy saving for 16V-77 °F despite of two point less voltage then 18V-68 °F or can we get electric energy saving in this condition? I hope I had explained properly two different conditions and I wonder your opinion ...
Alaattin Tuna [returning]- TURKEY Sakarya
December 5, 2020
A. Hi. I can't make it clearer, Alaattin: OF COURSE if you formerly required 18 V and you are now able to produce identical anodizing at 16 V and the same Amperage and the same anodizing time, you are saving a lot of energy both on your rectifier and on the heat input which your cooling system must remove from the bath. I keep confirming that I concur with the answer to that question again and again.
... but you seem to keep requiring that I accept the claim that the anodizing is identical, and then to accept that claim as proof that the conductivity must be 18/16 as high at 77 °F as it is at 68 °F. If it is, it is; but please just accept that we agree on the energy saving formula without demanding that I accept that the anodizing is identical and that the solution conductivity is that much higher at 77 °F than 68 °F 🙂
Here is a resistivity (conductivity) chart for 15% sulfuric acid at 68 °F and 77°F which shows higher conductivity, but not by that much.

from


Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
Sorry! Finishing.com is temporarily Read-Only.
Ted Mooney is retiring but I have several offers to take it over.
We're working hard to make sure we find it the best new home.
