| Search our quarter-million Q&As |
Home of the finishing HOTLINE since 1989
-----Restauranteur asks if stainless steel okay for lemon juice (citric acid)?
Q. I work for a restaurant chain that is switching from plastic cambros to all stainless steel. They seem to believe a myth that tomato acids react with stainless steel. I have already disproved this myth.
Now they are saying that our guacamole (which is made with a touch of lemon juice) will react to stainless steel and become discolored. I disproved the color theory yesterday. Now they are saying it is a slight taste difference. I think it taste different because they want it to and their mind is telling them it does. The whole reason behind the argument is that we are using plastic liners in the tomato products and guacamole pans. I am trying to eliminate the use and save the company money. The proof they want seems like they want me to prove there is no such thing as Bigfoot, when there is no proof that he actually exists.
Chris MilazzoRestaurant - Winter Park , Florida
2006
A. Hi Chris.
I think the source of that idea might be that aluminum (not stainless steel) can be sensitive to such acidic foods. Since many different product lines may use either metal, and they look vaguely similar, many people probably don't pay much attention to the difference :-)
Luck & Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
A. Chris, as a metallurgist with almost 25 years experience dealing with stainless steels, I'm in your corner. You'd better not serve anything that will attack stainless steel, as that substance will quickly and dangerously corrode the palates of your diners :) Nor can I believe any food or beverage will leach out metal from stainless, affecting the taste of the product.
That said, there are different stainless steel alloys. Pans, sinks, and some flatware are made from the 300 series, so-called "18-8" varieties, which have quite good corrosion resistance. The cutlery grades like 440C are used to make knives, because it gets hard and keeps an edge: yet its corrosion resistance is much less than the 300 series grades, and it might show rust if you left it in salt water for a time.
You've also got to be careful about scrubbing the stainless steel with a Brillo pad, or any other steel wool contact. If those plain steel fibers imbed in the stainless, they'll rust and take the surrounding stainless steel with it. Perhaps this is the source of your co-workers' concerns?
One last thought regarding the taste issue: I'd find it far easier to believe that plasticizers, dyes, UV inhibitors, and other additives to plastics can theoretically leach out into food or beverage, than metallic ions from stainless steel, especially as time and temperature cycles from cleaning march on.

Lee Gearhart
metallurgist - E. Aurora, New York
2006
A. I agree with Lee. As long as the stainless steel you are using is not contaminated with iron, ferric oxides, etc., and is not "rusting". The stainless will not change the color or taste.
To obtain the highest quality pure finish for stainless, have your parts electropolished. This will remove all contaminants and leave the surface ultra-clean. Also if you can, demand at least 304 or more preferably 316 stainless steel.
My suspicions are that your "tasters" have gotten used to the leached plastic taste in the food as opposed to the REAL taste they are missing if they were to use leach free food prep equipment.
stainless steel electropolishing shop - North Vancouver, British Columbia, Canada
September 18, 2009
![]() Appreciate the replies here! I stumbled upon this while googling "stainless steel" and "lemon", together, as I have a "Designed for Living" stainless steel water bottle, and was wondering whether my adding a couple of lemon wedges into my water would introduce any type of corrosive affect on the interior of the bottle.
Appreciate the replies here! I stumbled upon this while googling "stainless steel" and "lemon", together, as I have a "Designed for Living" stainless steel water bottle, and was wondering whether my adding a couple of lemon wedges into my water would introduce any type of corrosive affect on the interior of the bottle.
From the replies I see here (perhaps contingent on the grade/type of stainless), I shouldn't worry. Thank you.
- East Greenwich, Rhode Island USA
March 3, 2011
A. Of course, Stainless Steel Lemon Squeezer is safer than others. As my opinion, Stainless steel is best quality material for making any squeezer and it's also durable and long-lasting. Stainless Steel is very hard & smoother than other squeezers. It is easy to hold & very comfortable for the manual press.
Archer Richard- United Kingdom
May 24, 2018
|
|
Q. Lee, my daughter has low iron. I was thinking that lemon juice/water in a steel water bottle could help increase her iron levels. From your answer it seems no. Do you have any easy solutions for increasing her iron rather than taking a tablet? susan shaffer- newcastle [Australia] November 2, 2023 
A. Hi Susan. We are metallurgists & metal finishers, not nutritionists ... but if she likes Indian food the problem might be solved because Curry seems ideal as a source of iron. A. The simplest option I can think of is to have more red meat, next would be to use cast iron cookware, or carbon steel. Cast iron cookware is quite cheap and lasts near forever with a little bit of care. Andrew Speer- Ballarat, Australia November 21, 2023 |
Metallic taste in lemon curd from stainless pan?
Q. Yesterday, I made a lemon curd to top a cheesecake. The process involved cooking lemon juice and sugar together for several minutes. Instead of using a non-stick pan that I have used several times in the past, I used a stainless pan that is supposed to be non-reactive. When the cheesecake was tasted, I noted a metallic taste that was not present in earlier cheesecakes. Although stainless steel is non-reactive for most cooking applications, it is still a steel alloy. Will an acid such as hot lemon juice leach iron from stainless steel?
I'm thinking that I should look for a pyrex sauce pan for cooking acidic foods.
Cooking enthusiast - Orange, California, USA
March 20, 2011
A. Hi Ralph. Did you taste it with your fingers or use a fork? The only times that I personally have experienced a metallic taste is with silver plated flatware that is worn. The underlying nickel-silver substrate has a strong metallic taste if the plating is worn through in a spot. I don't think iron has a metallic taste.
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
[editor appended this entry to this thread which already addresses it in lieu of spawning a duplicative thread]
Q. I've been hooked on lemonade for awhile. I have to use plastic bottles to drink from because I have Multiple Sclerosis and I spill a lot. Today I purchased a stainless steel sports bottle from L.L. Bean because the plastic bottle becomes cloudy after a few refills (is the cloudiness from plastic dissolving or from the lemon bleaching?). Did I make a mistake? Will the stainless react with the lemonade?
Please answer quickly--I might have to cancel an order.
Dona
- SOMERS PT, New Jersey
January 22, 2012
A. Hi, Dona. It's a public forum; people may not see and respond to your posting for weeks or months, but we appended your inquiry to a thread that may already answer it to your satisfaction.
Fortunately, L.L. Bean has an unconditional money-back guarantee: "We do not want you to have anything from L.L. Bean that is not completely satisfactory", so if someone eventually convinces you that there is cause for worry, you can return it then :-)
Regards,

Ted Mooney, P.E.
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
Q. I got a "stainless steel" cup from Starbucks, but there is no mark on the cup indicating the "grade" of stainless being used. Does this mean it is actually aluminum? A magnet does not stick to it. I drink water with lemon oil every day. I have been told NOT to use plastic because it will break down petrochemicals. I have been using glass, but thought the tumbler I found would be a great alternative. How do I know what type metal is being used and if it is ok? Thank you.
S Cleveland- dallas, Texas usa
July 20, 2014
A. Hi, S. When you say it was "stainless steel", do you mean it looked something like stainless steel to you, or that it was actually advertised or labeled as stainless steel? I'm sure Starbucks wouldn't lie about such a thing. And I'd bet that they, like almost every other manufacturer, has an 800 number that you can call to find out. Stainless steel weighs nearly 3x what aluminum weighs, so you may be able to tell just from the heft, but if it's not a hollow thermos bottle you can surely easily tell from the Archimedes experiment. Good luck!
Regards,

Ted Mooney, P.E.
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
A. The stainless steel of type 316 is an austenitic chromium nickel stainless steel containing 2% molybdenum. this addition increases resistance to pitting, and provides increased strength at elevated temperatures. Corrosion resistance is improved particularly acidic medium such as sulfuric, hydrochloric, acetic, formic and tartaric acid.
I would guess that your problem would be solved using stainless steel of type 316 or 316L.
- gulbarga, Karnataka and India
July 15, 2015
A. While this doesn't directly pertain to acidic foods on stainless ware, it is an important thing to note in rare cases: Be careful about mixing different metals with foods. If you put tinfoil over a steel pan for instance, it may use the food as an electrolyte and create a crude battery (power cell technically.) This can greatly accelerate the reaction between pan and food and you may even see a blackish residue leeching from one of the metal surfaces into the food.
Matt Bower- Red Deer, AB, Canada
July 6, 2016
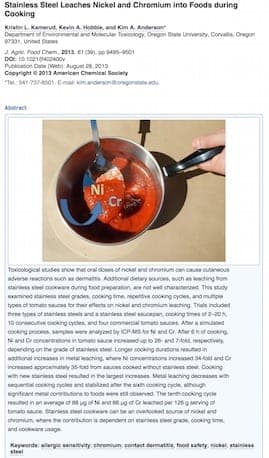
A. Two studies:
Doru Georgescu- Bucharest, Romania
September 7, 2016
Ed. note: Thanks Doru. Readers: the link for the second article is to the abstract only; the actual article is behind a pay wall
A. I just wanted to mention, with regard to the 2 studies posted in the last comment - The first PDF had some great information on corrosion; however, looking closely at the grade of stainless steel on which the study was done, I found "stainless steel grade 201" (bottom of page 3) as well as "Cr 16-18% / Ni 3.5 - 5.5" in the chart immediately following.
The concluded leaching rate in report #1 should not be compared to cooking grade stainless steel of 304 (18/8), 302 (18/10) or 316 (18/10 MO)(surgical grade) because the rate of corrosion in reported grade 201 is MUCH higher than what we normally use in our kitchens.
I have no comment about the second report because I will not be paying $40 for 48 hours of access to their website, but I can only assume that the posted excerpt of findings is along the same lines and has been gathered from a much lower grade stainless steel.
If anyone has similar reports on grades 302, 304 and 316, I invite them to post a link :)
- Las Vegas, Nevada
December 2, 2016
opinion! Hello C.
I won't pay $40 for 48 hours of access either! As a society we need to figure out a good way to compensate researchers for authoritative work, but charging for temporary access to technical info is reckless!
How does it feel knowing that your doctor no longer has access to the electronic textbooks rented to her while she was in school, so must choose between guessing from memory what they said or paying $40? How do we like driving over a bridge designed by an engineer in the same situation :-)
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
December 2016
P.S. June 2020: We can be heartened that negotiations between M.I.T. and Elsevier have broken down; my limited understanding is that M.I.T. will not let Elsevier continue to have that sort of control over the research they have done.
opinion! The fees asked to access academic papers is a disgrace and impedes the progress of knowledge.
However there is frequently free access by simply running a search on the full title. Hence
1. http://www.electrochemsci.org/papers/vol10/100503792.pdf
My main comment is that this refers entirely to type 201 stainless which I would not regard as a standard food grade.
2. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4284091/
I come from a time before the atomic absorption spectrophotometer
⇦ on
eBay
or
Amazon [affil link] . Since they arrived on the scene we have been inundated with more and more data on trace elements that are rarely significant outside academic circles.
As an old chemist with an interest in toxicology, I have no idea what my SS pots are made of and no intention to ask.
It is probable that the 'metallic' taste some people experience is due to an electrolytic cell set up between the cup and the body (metallic fillings would enhance the effect).

Geoff Smith
Hampshire, England
Ed. note: Geoff's 2nd URL does indeed work to offer free access to the 2nd article suggested by Doru
Q. I took lemonade to work in a stainless steel bottle. I got a stomach ache and suddenly had dry heaves and diarrhea - felt fine later . Could drinking lemonade out of stainless steel container have made me sick?
Robin LeFlore- Tioga, Texas, usa
November 28, 2016
A. Hi Robin. Although anything is possible, I'd suspect a long list of more probable things before considering that possibility, and I bet that someone with medical training would agree :-)
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
Q. I have a stainless steel (316) water container volume 90 lt.
My question is - how do you remove the metallic taste in the water. The container has been filled twice but the metallic taste remains.
- Cape Town South Africa
February 15, 2017
A. Billy,
If the stainless really is leeching iron into the water, just fill the container with a solution of at least 4 weight percent citric acid and let it soak for at least 30 minutes. (Or partially fill it and rotate the container so that all surfaces have at least 30 minutes exposure to the solution.) Rinse it out with water afterwards and let it dry in air overnight.

Ray Kremer
Stellar Solutions, Inc.
McHenry, Illinois

A. Here's an article about stainless steel grade 316 leaching nickel. It's free to read.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4284091/
- Metro Manila, Philippines
December 14, 2018
Ed. note: Thanks Mackenzie! This is the same URL that Geoff & Doru suggested. Great minds etc.
A. I am Robert Seals the inventor of Klean Kanteen which started the stainless vs plastic revolution; I sold the company years ago. I had the bottles tested back then because as a metal worker I was concerned about electropolishing, which I did not trust. Using an acid equivalent to lemon juice it was found to leach nickel over the acceptable amount allowed by FDA standards. I was concerned enough to print FOR WATER ONLY on the bottles as the same tests showed no detectable metal when water was used.
Electro polishing is not the way to passivate any food container. The old fashioned polishing method is by far is the best way to seal the metal but it's too labor intensive for production. The public is being duped on this one.
Robert SealsArtists - LOS GATOS California USA.
February 21, 2019
![]() Thanks Robert. Maybe stainless steel isn't quite as impervious to acidic food & beverage service as we thought? What grade of stainless did you use?
Thanks Robert. Maybe stainless steel isn't quite as impervious to acidic food & beverage service as we thought? What grade of stainless did you use?
But I'm not understanding your point about electropolishing at all, which I understand to be the only safe & accepted way to polish pharmaceutical and dairy equipment. Are you saying that you have found that mechanically polished surfaces leach less nickel than electropolished surfaces? If surfaces are mechanically polished, what passivation method do you feel is indicated?
Thanks again, although without any data I cannot concede that electropolishing leaches more than mechanical polishing.
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
A. It's an interesting topic. We have data showing that citric acid exposure does not remove chromium or nickel from the surface of stainless, in order to show that citric acid passivation has a lower waste disposal task than nitric acid passivation, which definitely removes some amount of chromium and nickel.
I rather suspect that even "highly acidic" foods are closer in acidity to a citric acid passivation bath than a nitric acid passivation bath. However, I would eagerly read any further studies performed using lab or real-world conditions.

Ray Kremer
Stellar Solutions, Inc.
McHenry, Illinois

February 25, 2019
Sorry! Finishing.com is temporarily Read-Only.
Ted Mooney is retiring but I have several offers to take it over.
We're working hard to make sure we find it the best new home.