
| Search our quarter-million Q&As |
Home of the finishing HOTLINE since 1989
-----Electricity from fruit (cont'd)
< Prev. page (You're on the last page of the thread)
Q. Hi:)
I did an experiment and found that limes, apples, grapefruit, mangoes and plums all conducted energy better than lemons and oranges, which I expected would conduct more since lemons are what everyone uses for their projects. I've done some research but everyone seems to have different opinions on the topic. What do you think makes some fruits conduct more energy than others?
Thanks a lot
- nyc, New York, U.S.A
May 11, 2015
![]() Hi Sophia. You didn't mention what grade you are in, but you need to be rather careful with terminology like "conducted energy better" and "conduct more energy". You are expressing yourself rather vaguely about some observations you made. Trying to answer "why" something happens that you haven't clearly enunciated, can confuse and mislead you :-)
Hi Sophia. You didn't mention what grade you are in, but you need to be rather careful with terminology like "conducted energy better" and "conduct more energy". You are expressing yourself rather vaguely about some observations you made. Trying to answer "why" something happens that you haven't clearly enunciated, can confuse and mislead you :-)
The role of the fruit juice in this case is probably mostly to provide a conductive electrolyte. That leads me to a thought: Have you ever eaten orange segments? Suppose you've peeled an orange, and you've eaten most of it, and now you have two segments left. Do you think you'll get the same or different results if you stick both of your probes into the same segment, versus sticking one probe into one segment and one into the other, being careful not to puncture the membranes separating the two segments?
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
May 2015
Q. Hi there. Regarding that project I had a similar one. Just that am in advanced level. We planned to charge a phone using the fruits. Is it possible? Need advice on how to do it.
Steve Paul- New-York. Usa
May 29, 2015
A. Hi Steve. Almost anything is possible, surely including charging a phone using fruit batteries...
What voltage does a phone charger for the phone in question provide? What amperage does it provide? Hint: my questions provide the advice you asked for on how to go about it.
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
Q. Hi,
My son (12) is doing a year 8 (not sure what that is in other countries) holiday science project. We are running a small digital clock from various produce and liquid, for example a Kumara (sweet potato), bread, water, etc. using copper and zinc electrodes attached to wires from clock to two of each item and a white wire with a copper and zinc electrode running to the opposite. One of the things he used which we found that did not run the clock but cannot find a reason why from Google is butter. Are you able to give us the reason why please? I was wondering if butter in fact acted as an insulator rather than a conductor. Another one we found was salt (two small cups). Thanks,
- New Zealand
July 7, 2015
A. Hi Sherryn. You're halfway there in finding that some things will support this battery action if the probes are inserted into them and some will not. But the issue or principal that your son should be getting is what is actually happening that accounts for the electricity flowing ...
If a single metal electrode is placed into acid it will slightly dissolve into the acid, much like salt or sugar will dissolve into water to a given equilibrium point (if you add too much salt or sugar it just settles on the bottom as a syrup). As the metal slightly dissolves into positively charged ions in the solution (atoms less one or more electrons) this leaves excess electrons on the metal electrode, and they attract ions back and reform metal, and this is what comprises the "equilibrium".
But if you put both copper and zinc rods into the same solution, when the dissolved copper ions (atoms of copper less two of their electrons) happen to reach the zinc electrode, they will steal two electrons from the zinc metal and become copper atoms again, plated out onto the surface of the zinc. Why copper has this stronger thirst/affinity for those electrons than zinc may be beyond your son's level, but the principal of what is happening probably isn't.
As mentioned, when the copper atom dissolved, it left those two electrons on the copper electrode, and they will now flow through the wiring to get over to the zinc electrode to balance things out. This flow of electrons through the wiring is what we call electricity. The reaction will continue until the zinc is completely covered with copper and the surface of the two electrodes is the same metal, so nothing further can happen and the "battery" is dead.
Butter is oil rather than water, and will not support the dissolving of metal into ions. So it's not that it's an insulator, it's that metal can't ionize into it. Similarly, dry salt crystals cannot support metal ionizing into it. Good luck.
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
July 2015
Q. Actually, I have four questions:
1). What does zinc look like?
2). Do I need a high watt battery to power this circuit, and how many do I need
3). Can I also use an unripe orange?
4). And is it a 60 watt bulb light I will use to the circuit or just a smaller one?
- Lagos,Nigeria
October 15, 2015
A. Hi Ignatius.
1. Zinc is gray and metallic looking. Galvanized nails, galvanized roofs, and boat anodes all have a zinc surface.
2. I don't know what circuit you are speaking of, but I have been speaking of a "fruit powered battery", where a piece of copper and a piece of zinc stuck into the fruit comprise the battery.
3. Yes, you can use an unripe orange.
4. Your circuit will be unable to light even the tiniest penlight flashlight bulb. You would probably need a dozen fruit batteries wires in parallel to light that penlight battery, or a few to light even a small LED per the youtube video we referenced.
Luck & Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
October 2015
Q. How does the amount of acidity in a fruit affect the amount of electricity it can produce?
Iliyan Kajani- Atlanta, Georgia, U.S.A.
January 4, 2016
A. Hi Iliyan. That question was answered in detail several times on this page already. Please try to read and understand the explanations, then to re-phrase your question so we can resolve any remaining uncertainty you have.
Also, try to think carefully what the word "amount" means to you because I can think of at least two different meanings (alkalinity and pH) related to "the amount of acidity", and many meanings (voltage, current, power, energy) regarding electricity. Good luck.
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
January 2016
January 12, 2016
My 5th grade son is conducting an experiment where he wants to find out if changing the state of the potato will influence its electrical output.
I want to make sure that he is conducting the experiment correctly -- Would appreciate your help.
He tested a potato battery made up of 3 yukon gold potatoes cut in halves and was able to light a 2 Volt small LED light.
He now wants to boil 3 potatoes and test these to see if the voltage is higher when the potato is boiled,
He then wants to place 3 potatoes in salt water (1 tablespoon of salt dissolved in 1 cup of water) and test the voltage
Finally he will place the 3 potatoes (cut in half) on salt for a day and test the voltage.
HE is hoping that by changing the state of the electrodes he will produce a larger flow path and therefore allow more electrical output to generate when using the potato.
His hypothesis is that the potatoes placed directly on salt will give the most power as it will increase the electrodes the most.
Is he on the right path? Is he stating his question correctly?
Thank you for any help with this
- Fernandina Beach, Florida, USA
A. Hi Paola. The basic problem is that it's the two metals which generate the voltage, not the potato. All the potato does is provide a conductive liquid path. But I would guess that, yes, if you use a strong salt solution, with the metals placed close together, the resistance of the circuit is lower and you should be able to get more current to flow, and the LED to glow more brightly. With more current flowing, the battery will go dead sooner though.
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
Q. Hello, I'm creating electricity in my 7th period class. We are trying to create electricity from an apple, orange, banana, and a potato. I was wondering if you can actually do this and does it actually light up an LED light?
emili quam- devils lake North Dakota
February 10, 2016
A. Hi Emili. Please look at the youtube video of the lemon battery on this page. The person making the video found that he had to wire 4 lemon batteries in series to light a small LED.
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
February 2016
Q. Hello!!
So my 6 year old is participating in his first Science fair! We both decided to do a Digital Fruit Clock! (He got a "Lemon Clock" kit for Xmas complete with two copper and Zinc rods, red wire, Black Wire, white wire, and a digital clock) We started to do experimenting on fruits and NON fruits. The clock worked with an Avocado, Lemon, Banana, and a pear. The Clock did NOT work with Peanut butter, Butter, and a chocolate chip cookie. We were trying to find things it WOULD NOT work with and found that water works, milk works even BREAD! Why Bread? In conclusion we need to figure out WHY it works for fruit as opposed to butter, Chocolate chip cookie, and peanut butter. What is it that the Milk, water, bread and fruit have in common that it would make the clock work but NOT the butter, Peanut butter, and cookie... PLEASE HELP!!!
- Seattle, Washington
March 6, 2016
A. Hi Kimberly. I hope you tried these materials repeatedly because it would be foolish for him to carefully explain why something is true if it's not true :-)
But assuming the results are repeatable, it means some of the materials conduct sufficient electricity to run the clock (it probably takes very very little) and some do not. Anything with a little water in it probably works, and things that are dry, or based on oil rather than water, do not. If you happen to have an electrical meter you could measure the resistance of each of these objects.
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
March 2016
Q. Hi, I have a science fair project at school. And I chose "Which combination of vegetable and fruit has the highest voltage?" And it's like a different thing to do here.
Brokson Kevin- Mandalay, Myanmar
March 15, 2016
A. Hi Brokson. Sorry, but it's not the fruit or vegetable which you choose that determines the voltage, but the two metals. I'm not happy to have to tell you that, but it is what it is :-)
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
Q. Hi, Please help me a year 6 student get an answer for why energy is increased in voltage if more fruits are used?
khushi bajaria- mombasa,ganjoni,kenya.
April 5, 2016
A. Hello Kushi. Copper has a much stronger pull on electrons than zinc has: about 1.0 volts worth. So if you put a piece of copper and a piece of zinc into a fruit, a meter would record about 1.0 volts between them as some of the electrons in the zinc metal are pulled through you wiring and over to the copper metal. If you put two such batteries in series (connecting the positive end of one to the negative end of the other, much as you stack 1-1/2 volt batteries in various electronic devices, you would get about 2.0 volts between them. If you stack 4 of these 1.0 volt batteries you get about 4.0 volts.
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
April 2016
Q. Can you make a banana battery?
Sharnujan Sivanethura- Toronto, Ontario, Canada
April 14, 2016
A. Using any fruit with a high acid content and 2 Dissimilar metals will generate electric current. Peanut butter is not terribly acidic so you won't get much if any electricity. You can build a simple "Voltaic Pile" (named after Alessandro Volta. Also where we derive the name "Volt" from). All you need are some silver coins and copper coins, some vinegar
⇦ in bulk on
eBay
or
Amazon [affil link] and blotting paper. Simply cut some squares of the blotting paper slightly larger than the coins and moisten them with the vinegar. Place a copper coin on the table, put a piece of the blotting paper on top then put a silver coin on top of that. Keep repeating the process making sure to alternate the coins. When you have your desired "stack height", touch your multimeter probes on the top and bottom coins and be amazed! (hopefully)
Chuck, Melbourne Australia
Tube Bending, Powder coating and Metal Fabrication - Thomastown, Victoria, Australia
May 31, 2016
![]() It's okay to build the stack high enough that you're amazed; but don't build it so high that you're shocked :-)
It's okay to build the stack high enough that you're amazed; but don't build it so high that you're shocked :-)
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
Q. Hello Ted,
I've seen many reports online of a boiled potato producing 10x more electricity when used in these typical zinc-copper "fruit batteries" than a raw potato, but these reports seem to all be reiterations of the same news report from 2010 about the Hebrew University of Jerusalem. I can't find anyone else actually doing an experiment using a raw and a boiled potato and documenting it online (outside of the recent question this year from Paola here on this thread, but her child's results weren't posted). Here is an example of this finding: http://www.smithsonianmag.com/innovation/a-potato-battery-can-light-up-a-room-for-over-a-month-180948260
Based on all of the wonderful information you've provided here, I have my doubts that this will make a difference -- certainly that much of one, especially since you've pointed out that there is a maximum which can be produced using zinc and copper as electrodes. Even in ideal conditions. Unfortunately, I don't have time or resources to create the experiment myself, as I'm just doing research for a paper (we don't even have pennies in this country).
Can you please tell me if you think it's possible to produce exponentially more voltage in the potato battery setup by using a boiled one? How can that texture make a difference? Cheers in advance!
- Katoomba, NSW, Australia
July 3, 2016
We'd love to give credit for this graphic explaining Ohm's Law (A = V / Ω) but we see it on many websites with no hint of whose work it actually was :-(
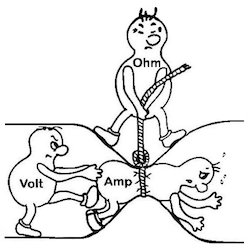
Hello Jemima. The voltage of the battery will remain unchanged because it is a function of the two metals employed (copper and zinc). But the maximum current that metal plates of a given size can produce is increased because the boiled potato is a better conductor. The practical implication is that more power can be generated in a given space, i.e., it's a more practical packaging method/design for the battery.
The biggest problem is that this corrosion of zinc and copper isn't really "free" energy because it costs more energy to make zinc and copper from ore, i.e., to un-corrode them, than you can get from corroding them like this. Batteries can be a convenient storage device for energy, but they don't actually produce energy, they just release a portion of the energy that was stored in them.
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
July 2016
![]() Hi -- just a quick note to thank you so much for taking the time to provide such a thoughtful and articulate answer to my boiled potato question, and so quickly! It's really wonderful that you've been keeping this thread and information going for so long. Cheers from Oz!
Hi -- just a quick note to thank you so much for taking the time to provide such a thoughtful and articulate answer to my boiled potato question, and so quickly! It's really wonderful that you've been keeping this thread and information going for so long. Cheers from Oz!
- Katoomba, NSW Australia
July 3, 2016
Q. I'm doing a project for the equivalent of a science fair. I'm thinking about doing something with fruit conductivity, but one question has piqued my interest. Why must zinc and copper be used to make a fruit part of an electrical circuit?
thanks in advance,
Edward, a year seven sudent.
- Sydney, New South Wales, Australia
August 9, 2016
A. Hi Edward. For reasons that are beyond grade 7 level and maybe my level, zinc attracts electrons more strongly than copper does. So if you put the two metals into a conductive solution but not touching each other, and you attach a wire between them, electrons will flow through the wire from the copper to the zinc. As this happens, the copper atoms on the surface of the copper, having lost their electrons, do not remain a metallic solid, but dissolve as positively charged copper ions into the solution (much like salt or sugar dissolve into water). The positively charged copper ions migrate through the solution until they reach the zinc electrode and are able to regain their electrons and come out of solution, as copper plates onto the zinc electrode. The process of electrons flowing through the wire from the copper to the zinc, and copper ions dissolving into solution and traveling to the zinc electrode continues until the zinc is completely covered with copper and the process dies out.
You do not have to use zinc and copper, you can use any two metals as long as one has a stronger attraction for electrons than the other. You may have heard of carbon-zinc batteries, nickel-cadmium batteries, etc. But zinc and copper are readily available, relatively cheap, and the difference in attraction of electrons is large, so they generate a good voltage (about 1.1 volts).

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
Q. Hey, How can I measure the moisture content of a fruit by passing an electric current through it? And how much current should be applied.
Emmanuel Mwadime- manila, philippines
January 5, 2017
A. Hey, Emmanuel, I don't think you can. What leads you to believe that it can be done?
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
January 2017
January 16, 2017
And what unit of measure are you looking for "moisture content" in?
Electrical conductivity in this context requires there to be liquid, and for that liquid to have ions in it. The liquid is usually taken as a given, so conductivity is often used as a measure of how many ions there are. If the liquid dries up and the ions turn into solid salt, it would pretty much lose conductivity, but it's doubtful you would need to measure the current to be able to notice that.

Ray Kremer
Stellar Solutions, Inc.
McHenry, Illinois

! I'm making a science project about what kind of vegetable produces the most power.
david hardcore- colorado
September 12, 2017
September 2017
![]() Hi David. Good to hear. Let us know what you find out about that. Thanks.
Hi David. Good to hear. Let us know what you find out about that. Thanks.
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
Q. My daughter a fourth grader has chosen to conduct an experiment where she will test fruits and vegetables to see if they are good conductors of electricity and which fruits or vegetable conducts the best. She understands independent and dependent variables and constant but I am having a difficult time making her understand control in her experiment - P.S. I am starting to get a little confused too. Does every experiment always have a control?
Thanks
Tenzing
- Davenport, Florida, USA
October 6, 2019
A. Hi Tenzing. No, every experiment does not require a control. A control is used for "what would have happened if I didn't add chemical 'X', or do procedure 'Y', or how do I account for the presence of 'Z'?".
Imagine that your daughter's teacher says "Iodine might be a terrific conductor of electricity. Please prove/disprove it by measuring the conductivity of a beaker of tap water and then, because pure iodine is a bit hard to get and so very little is needed, add iodized salt to a 2nd beaker of water and measure its conductivity". It would probably immediately occur to you that the salt rather than the iodine might explain the increase in conductivity. In this case you might decide to add non-iodized salt to a 3rd beaker of water and measure its conductivity. That 3rd beaker is your control -- it lets you determine whether the tiny amount of iodine really explained the increased conductivity, or whether the salt you inadvertently added to get the iodine in there was the important factor in the increased conductivity.
Sometimes the 'control' is implicit or understood: When you measure the conduction of the fruit or vegetable with the meter leads say 1" apart, an implicit or understood control is that when the meter leads were 1" apart in air the conductivity was for practical purposes zero, so the leads being in the fruit is what produced the conductivity. Good luck.
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
October 2019
October 12, 2019
Q. Thank you so much for answering my question. It is very precise and simple.
I have another question. I see and read so many articles about fruits and electricity and vegetables and electricity. I understand that using two different wires will make a battery but we want to use two of the same wires and try and find out which fruits and vegetables are better conductors of electricity. I understand we need a multimeter to measure (am I correct in using a multimeter) the voltage and two of the same wires.
The question is: 1. will using two zinc or two copper wires give different results.
2. I am still confused between measuring conductivity and producing electricity. Our goal in this experiment is to measure conductivity and not generate electricity so do we measure resistance or voltage.
3. Every experiment or article we look at on fruits and vegetables conducting electricity, it always goes to a vegetable battery or a lime battery. We don't want to make a vegetable or fruit battery, just need to try and look at vegetables and fruits as to which is the best conductor of electricity, please clarify.
4. Conducting is not the same as generating, am I correct? Please explain the difference in a way that a fourth grader can understand
Your help is much appreciated
- Davenport, Florida
A. Hi again. Yes, you need a multimeter, and you need to measure resistance. We probably need to understand exactly what resistance, conductance, resistivity, and conductivity mean. The cute little graphic further up the page shows the relationship between voltage, current, and resistance. Voltage is measured in volts, current is measured in amperes (amps), resistance is measured in ohms. If you know two of the three, the third is easily solved for from the relationship Amps = Volts/Ohms. Conductance is the reciprocal of resistance, and is expressed in mhos; mho doesn't really mean anything, it's just ohm spelled backwards as a reminder. If the resistance of something is 10 ohms, its conductance is 1/10 mho; if it's resistance is 1/20 ohm, its conductance is 20 mho.
Resistance and conductance are a measure of an entire system: the resistance read on the meter is the resistance of the copper or zinc wires plus the fruit of vegetable. The way it works is the multimeter has a battery in it and when you set it to ohms or resistance the battery is used; it produces a voltage, say 1-1/2 volts, which runs through the wires and the fruit and the meter needle ... the amount of current that can flow through the system against this resistance causes the meter needle to move, and the scale of ohms is painted under the different possible needle positions.
You perhaps shouldn't even use the words resistivity and conductivity, but they are properties of a material rather than a measurement of a system. Rubber is a very high resistivity, low conductivity material. And copper is a very low resistance, high conductivity material. But even with a low resistivity material like copper wire, if you make the wire 100X as long its resistance will be 100X as high.
In practice you are going to touch your meter leads together and note that the resistance is essentially zero. That is your 'control' that accounts for the wires so you are measuring just the resistance of the fruit. Then you are going to put the meter leads into the fruit or attach them to pennies or copper wires you put in the fruit. You should always place the leads or wires approximately the same distance apart in each fruit because, as mentioned, resistance increases proportionally to distance.
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
October 2019
Q. Thank you very much. You have been very helpful. My final question:
at what range do we measure resistance and once we measure resistance do we need to convert that into ohms?
Thank you :)
- davenport, florida, usa
October 17, 2019
A. Hi. If you use a digital meter it's probably "auto-ranging" -- it may shift the range up or down by itself and just give you the answer in ohms or megaohms (millions of ohms). If you use an analog meter (one with a needle) it's more fun and a better learning experience. Pick whatever resistance range you wish, touch the two leads together and 'zero' it if necessary. Then attach to your two probes in the fruit. If it reads very close to zero, try to move down to a lower range so the needle is nearer the middle; if it reads close to infinity, try to move up to a higher range so it reads nearer the middle. Resistance is measured in "ohms", so no conversion is necessary -- the meter reads in ohms. However, you may find that the meter range says 'X1000" or "X100,000" or "Megaohms" (X1,000,000), so you have to multiply what the needle says by that factor.
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
October 2019
Q. Thank you as always.
We got a multimeter, didn't mean to but it was an analog and it was quite a challenge, not as easy as the digital I presume. It had only two range for the resistance measurement, we stuck to the X1K range and measured each fruit and vegetable three times. Once with the copper wires two inches apart and twice with the copper wires one inch apart. Results validate that distance is proportional to resistance (at least we got that part correct). Measurement at the X10 generated no results.
Coming to the question : resistance in Ohms. We know that resistance is reciprocal to conductivity. So apple measured at (wires one inch apart at X1K) 15 ohms the first time and 17 ohms the second time so we took the average of 16 ohms, which is times 1000 and is equal to 16000 ohms. so conductance is 1/16000 ohms^mhos, are we correct? The average measurement of resistance of grapefruit was 22.5 ohms so multiply by 1000 we get 22500 ohms, so for grapefruit, conductance is 1/22500 [mhos] which is close to 0. So essentially higher resistance means lower conductivity ...
Please help we are CONFUSED :(
- Davenport, Florida, USA
October 21, 2019
October 2019
A. Hi again. Unfortunately this lesson seems beyond 4th grade level and your daughter may have little real concept of anything even after all your efforts. The main issue is that resistance and conductivity have no meaning except as a relationship between voltage and current, neither of which she has been introduced to (it seems from here), and neither of which she has any concept of (it seems from here). It's no wonder it confuses you, let alone a 4th-grader :-)
But maybe some things can be made just a bit clearer regardless. First, to be a stickler, I must note that you did not read 15 ohms or 17 ohms or 22.5 ohms so you must not word it that way, so I crossed it through. You were on a scale that read thousands of ohms, so you read 15,000 ohms, 17,000 ohms, and 22,500 ohms. You understand that, but it's also important to only say it that way. Secondly, everything is relative, and you mustn't say that 1/22500 is "close to zero"; small resistors in electronic circuits often have a resistance a thousand times greater than your fruits, and therefore a conductance 1000X smaller, and it's still not close to zero :-)
Lastly, the units we use in describing conductance is mhos, not ohms, which I'll now try to explain...
Back to what I alluded to earlier, resistance is a relationship between voltage (Volts) and current (Amps): that relationship is R = V/A.
So an "Ohm" is also or actually a Volt per Amp. We could say the resistance of your grapefruit was 22,500 Ohms or we could say that resistance was 22,500 Volts per Amp.
Conductance, as you realize, is the reciprocal of resistance: so C = A/V.
Thus, conductance is measured in Amps per Volt rather than Volts per Amp, and since Ohms means Volts per Amp we can't use that unit of measure to describe conductance, we must use mhos. Excellent work even if it wasn't fully understandable!
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
October 22, 2019
Q. It's been quite a ride - a journey of learning but we are left with more questions :)
We proved that resistance increases proportionally to distance. With the copper wires 2 inches apart we recorded for example apple showed 19 ohms which on a scale of thousand is 19000 ohms and with the wires placed 1 inch apart it recorded 15000 in the first trail and 17000 in the second trail.
Now my question is: the fruits or vegetable with the highest resistance has the worst conductance and vice versa. If that's the case we have some interesting results :).This is to make it easier for her report as we really need to get to the conclusion. Am I correct in saying that?
Conductance is reciprocal of resistance and measured in mhos.
We are having a difficult time wrapping up - her hypothesis that the vegetable with the most water in it will be the best conductor and grapefruit with its high acidity will be the best conductor among fruits needs to be proved or falsified. We have all the measurements for resistance - is it correct to simply take the reciprocal of each measurement and express it as the conductance for that fruit or vegetable.
Thank you very very much - you have been put science teacher through this entire project.
My daughter sends her best.
- Davenport, Florida, USA
A. Hi again. It is correct to say that conductance is the reciprocal of resistance and to express it in mhos.
Her conclusions are that apples are more conductive than grapefruits, and conductance decreases when the probes are further apart. She has disproven her hypothesis that fruits or vegetable with the most water and the highest acidity are more conductive: because the data she recorded contradicts that hypothesis, and all it takes is one example to show that the hypothesis is demonstrated to be incorrect. However, she should not go too far and claim that fruits & vegetables with less water or lower acidity are more conductive because a single example does not prove the general case.
Regards,

Ted Mooney, P.E. RET
Striving to live Aloha
finishing.com - Pine Beach, New Jersey
Need quick confidential answers? $25
Need project assistance? $100/hr.
October 2019
Q. This is going to sound ridiculous but her conclusion requires her to list some industries that will benefit from her experiment. I don't understand how this is applicable to a fourth grader but we have to follow guidelines and complete it. In the beginning she mentioned that the fact that fruits and vegetables can conduct electricity might make us want to look at other means or ways of making electricity or power. I understand that fruits and vegetables by themselves don't generate electricity but given the proper internal structure and high levels of ions super conductive ions in them, they can help to create a working current. Are we justified in saying that her tiny experiment will benefit the power industry :)
Tenzing Dolma [returning]- Davenport, Florida, USA
October 29, 2019
Sorry! Finishing.com is temporarily Read-Only.
Ted Mooney is retiring but I have several offers to take it over.
We're working hard to make sure we find it the best new home.